 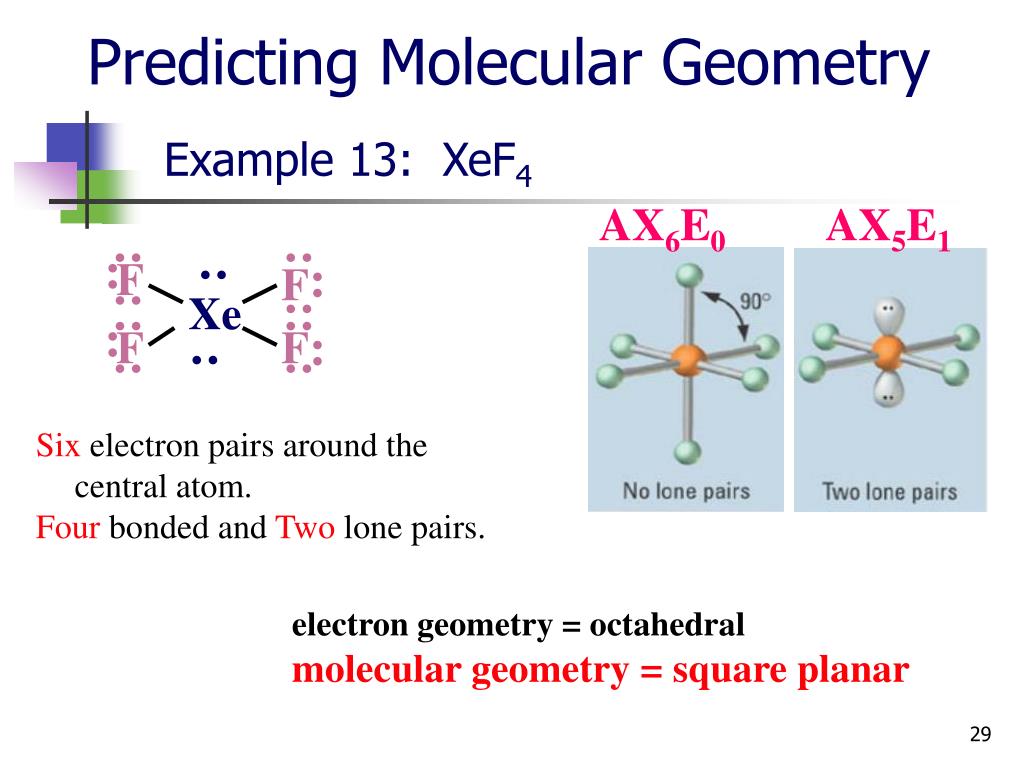
XeF4 is a nonpolar molecule so that its dipole moment is 0D All the polar molecules have a net dipole moment and all The dipole moment is the major aspect for any compound to be It is given as,ĭipole moment = Charge (Q) * distance of separation (d) The dipole moment is defined as the products of induced chargeĪnd distance of separation between the atoms. Have an MX4E2 structure which is according to VSEPR theory, an octahedralĮlectronic geometry means XeF4 has square planar molecular geometry.Īnd due to the symmetrical structure, the polarity of the Xe-F bond cancel each other and no dipole charge induced on the molecule. Localized in the form of 8 as Xe-F bonding and 4 as non-bonding pairs. The central atom of XeF4 (Xe) has a total of 12 electrons (4.0) and Xe (2.6) is 4.0-2.6= 1.4 which shows the Xe-F bond is polar according to In the XeF4 compound, the electronegativity difference between fluorine Periodic table, the fluorine (F) atom has the highest electronegativity value of 4.0, and It means higher the electronegativityĭifference between atoms, the more uneven sharing of electrons takes place. The atoms having higher electronegativity value attract theĮlectrons pair from the bonding partners. All the atoms have their ownĮlectronegativity value which varies from atom to atom. These are some of the major factors which are used to determine the polarity of the compounds: Electronegativity differenceĮlectronegativity is a kind of force exerted by atoms on theirīonding partners at the time of bond formation. Presence on the molecule which makes XeF4 a nonpolar molecule.Ĭheck Out: Is XeF2 Polar or Nonpolar? XeF4 Polar or Nonpolar (Based on polarity determining Non-bonding electrons that are presence on the central Xe atom which are aligned at 180 ° so that their effects are also nullified. 
The molecule is symmetrical so that all the Xe-F bondĭipoles are opposing each other, as a result, the net dipole charge becomes zero.īut as shown in the above diagram, there are still two pairs of the angle between F-Xe-F is 90 ° (Symmetrical). Octagonal electronic geometry which makes XeF4 molecular geometry square planar Is a vector quantity which means net polarity is the vector sum of individualĪccording to the VSEPR theory, MX4E2 structured have The Xe-F bonds are polar as the electronegativity differenceīetween xenon (2.6) and fluorine (4.0) is 1.4. Also, there are two lone pair of nonbonding electrons are In XeF4, there are four fluorine atoms are bonded with a central xenon atom. The same things apply here which makes XeF4 a molecule. Molecule to be polar or nonpolar in the above definitions of a polar molecule and Why is XeF4 a nonpolar molecule? Īs we have already known there are certain conditions for a The polarity induced on four Xe-F bonds are cancel each other and the net dipole moment becomes zero. This isīecause XeF4 has an octahedral symmetric geometry. Molecule despite four individual Xe-F bonds are polar. Now, I s XeF4 polar or nonpolar? XeF4 is a nonpolar Solvents like carbon tetrachloride (CCl4), carbon dioxide (CO2), benzene (C6H6) Water (called hydrophobic) and other polar solvents but are soluble in nonpolar Have symmetrical structure so that the induced partial charges on the individualĪtoms nullify each other and the net dipole moment becomes zero.Īs these are a pure covalent bond so they are insoluble in One more case for the nonpolar molecule if the molecules It forms by equal sharing of electrons between the atoms in a compound and this Nonpolar molecules are considered as pure covalent bond because They are soluble in water (called hydrophilic) and other polar solvents like hydrochloric acid (HCl), ammonia (NH3), Nitrogen dioxide (NO2)sulfur dioxide (SO2), etc. Polar molecules have polar covalent bonds or ionic bonds. Presence of lone pair of electrons or electronegativity difference between Moment and this is possible only if there is an electronegativity differenceīetween the associated atoms as well as asymmetrical geometry so that the inducedĬertain conditions for the molecule to be polar i.e. 
Polar molecules are those molecules that have a net dipole Have some ideas about what polar and nonpolar molecules are: Polar Molecules Whether it is a polar or nonpolar molecule in full detail. In this article, we are going to know about the polarity of XeF4 means It is soluble in water and hydrolyses at low temperature toįorm different compounds like xenon, oxygen, xenon trioxide (XeO3), and hydrofluoric acid (HF). Has density 4.040 g/cm3, molar mass 207.283 g/mol, and melting point 117 ☌. Xenon tetrafluoride is a white crystalline solid in appearance 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
